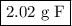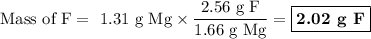Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 10:00
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3
You know the right answer?
How much fluorine (in grams) did the second sample produce? ? upon decomposition, one sample of mag...
Questions




Mathematics, 29.08.2019 00:30

History, 29.08.2019 00:30


Business, 29.08.2019 00:30


Mathematics, 29.08.2019 00:30

Mathematics, 29.08.2019 00:30

Mathematics, 29.08.2019 00:30





Mathematics, 29.08.2019 00:30


Mathematics, 29.08.2019 00:30


History, 29.08.2019 00:30