Answers: 3


Another question on Physics

Physics, 21.06.2019 23:50
Any color picture tube and electric field exerts a net force of magnitude 1.68x 10^-13 n on an electron the rest mass of an electron is 9.11 x 10^-13 what is the electron acceleration?
Answers: 3

Physics, 22.06.2019 00:20
Consider the particle-in-a-box problem in 1d. a particle with mass m is confined to move freely between two hard walls situated at x = 0 and x = l. the potential energy function is given as (a) describe the boundary conditions that must be satisfied by the wavefunctions ψ(x) (such as energy eigenfunctions). (b) solve the schr¨odinger’s equation and by using the boundary conditions of part (a) find all energy eigenfunctions, ψn(x), and the corresponding energies, en. (c) what are the allowed values of the quantum number n above? how did you decide on that? (d) what is the de broglie wavelength for the ground state? (e) sketch a plot of the lowest 3 levels’ wavefunctions (ψn(x) vs x). don’t forget to mark the positions of the walls on the graphs. (f) in a transition between the energy levels above, which transition produces the longest wavelength λ for the emitted photon? what is the corresponding wavele
Answers: 1

Physics, 22.06.2019 02:10
Astudent is performing an experiment comparing sound and light waves. the student gathers the following data. what conclusion does the student most likely make based on this data? light waves always travel the same speed; however, the speed of sound is determined by the medium that it travels through. all sound waves always have the same energy, so the temperature of the medium does not affect wave speed. light needs to vibrate particles, so it travels fastest in tightly packed solids, while sound does not need a medium, so it travels fastest in a gas. tightly packed particles in solids slow down the light waves; however, sound waves make particles bounce into each other, so they travel faster in solids.
Answers: 3

Physics, 22.06.2019 05:50
Acylinder with a movable piston contains 11.7 moles of a monatomic ideal gas at a pressure of 1.32×10^5 pa. the gas is initially at a temperature of 300 k. an electric heater adds 43200 j of energy into the gas while the piston moves in such a way that the pressure remains constant. cp=20.79 j k^−1 mol^−1 for a monatomic ideal gas, and that the number of gas molecules is equal to avogadro's number (6.022×10^23) times the number of moles of the gas. (a) what is the temperature of the gas after the energy is added? (b) what is the change in volume of the gas? (c) how much work is done by the gas during this process?
Answers: 3
You know the right answer?
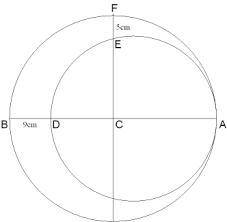
These lines are used to represent symmetry and paths of motion and to mark the centers of circles an...
Questions

English, 05.05.2020 14:14

Mathematics, 05.05.2020 14:14

Biology, 05.05.2020 14:14

Physics, 05.05.2020 14:14

English, 05.05.2020 14:14


Mathematics, 05.05.2020 14:14



Biology, 05.05.2020 14:14


Mathematics, 05.05.2020 14:14

Mathematics, 05.05.2020 14:14

Mathematics, 05.05.2020 14:14


Mathematics, 05.05.2020 14:14

Mathematics, 05.05.2020 14:14



Geography, 05.05.2020 14:14