Chemistry, 25.07.2019 10:30 jess7kids98
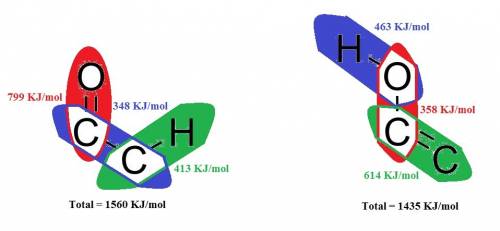
For most compounds with a single keto group in the molecule, equilibrium favors the keto form over the enol form of the compound. this is due largely to what?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 20:30
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1
You know the right answer?
For most compounds with a single keto group in the molecule, equilibrium favors the keto form over t...
Questions



Biology, 05.05.2021 21:50

Mathematics, 05.05.2021 21:50

Physics, 05.05.2021 21:50

Mathematics, 05.05.2021 21:50


Mathematics, 05.05.2021 21:50

Chemistry, 05.05.2021 21:50


SAT, 05.05.2021 21:50

History, 05.05.2021 21:50




Mathematics, 05.05.2021 21:50

Chemistry, 05.05.2021 21:50

Social Studies, 05.05.2021 21:50

Mathematics, 05.05.2021 21:50

Physics, 05.05.2021 21:50