Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2


Chemistry, 23.06.2019 08:00
The goal of this experiment was to answer the question "what is the effect of a gas' temperature on its volume? " you formulated the hypothesis below. hypothesis: if a fixed amount of gas is heated, then the volume will increase because the heat will cause the molecules of gas to move faster and further apart. to test this hypothesis, you changed the of the gas between 0 and 100°c (273 and 373 k) and calculated the resulting of the gas.
Answers: 2
You know the right answer?
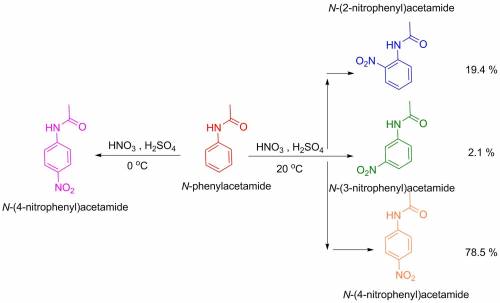
During the addition of no2+ to the dissolved acetanilide, the temperature must be kept cold. what pr...
Questions


Social Studies, 31.07.2021 20:10

Chemistry, 31.07.2021 20:10



Social Studies, 31.07.2021 20:10

Physics, 31.07.2021 20:10


English, 31.07.2021 20:10


Chemistry, 31.07.2021 20:10



Computers and Technology, 31.07.2021 20:10

Mathematics, 31.07.2021 20:10

Mathematics, 31.07.2021 20:10

Mathematics, 31.07.2021 20:10



Social Studies, 31.07.2021 20:10