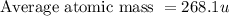Chemistry, 16.07.2019 00:10 aprilpendergrass
Anewly discovered element, y, has two naturally occurring isotopes. 87.8 percent of the sample is an isotope with a mass of 267.8 u, and 12.2 percent of the sample is an isotope with a mass of 269.9 u. what is the weighted average atomic mass for this element? 267.9 u 268.1 u 268.9 u 269.1 u

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Dwayne filled a small balloon with air at 298.5 k. he put the balloon into a bucket of water, and the water level in the bucket increased by 0.54 liter. if dwayne puts the balloon into a bucket of ice water at 273.15 k and waits for the air inside the balloon come to the same temperature, what will the volume of the balloon be? assume the pressure inside the balloon doesn’t change. type the correct answer in the box. express your answer to the correct number of significant figures. the volume of the balloon at 273.15 k is liters.
Answers: 2

Chemistry, 21.06.2019 23:00
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2

Chemistry, 22.06.2019 03:00
Select all that apply. a beta particle: is electromagnetic energy is an electron has zero charge is emitted from the nucleus has a +2 charge has a -1 charge
Answers: 1

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1
You know the right answer?
Anewly discovered element, y, has two naturally occurring isotopes. 87.8 percent of the sample is an...
Questions








Computers and Technology, 28.12.2019 07:31


Computers and Technology, 28.12.2019 07:31

Computers and Technology, 28.12.2019 07:31



Computers and Technology, 28.12.2019 07:31






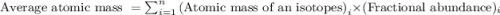
![\text{Average atomic mass }=[(267.8\times 0.878)+(269.9\times 0.122)]](/tpl/images/0094/3451/ba97e.png)