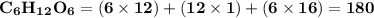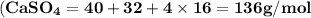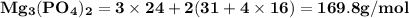Use the periodic table to calculate the molar mass of each compound below. give your answer to the correct number of significant figures. sodium hydroxide (naoh): 2 g/mol water (h2o): 4 g/mol glucose (c6h12o6): 2 g/mol calcium sulfate (caso4): 1 g/mol magnesium phosphate (mg3(po4)2): 1 g/mol

Answers: 2


Another question on Chemistry



Chemistry, 23.06.2019 01:30
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Use the periodic table to calculate the molar mass of each compound below. give your answer to the c...
Questions

Geography, 01.07.2019 00:00





Mathematics, 01.07.2019 00:00


History, 01.07.2019 00:00



Business, 01.07.2019 00:00


Mathematics, 01.07.2019 00:00





Biology, 01.07.2019 00:00

Social Studies, 01.07.2019 00:00

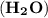 = (2 x 1) + 16 = 18 g/mol
= (2 x 1) + 16 = 18 g/mol