Chemistry, 17.06.2021 21:00 alaina3792
A chemistry student needs of carbon tetrachloride for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of carbon tetrachloride is . Calculate the volume of carbon tetrachloride the student should pour out. Be sure your answer has the correct number of significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Complete the following reactions using word and balanced equations including states. dilute phosphoric acid is added with a calcium hydroxide solution.
Answers: 1

Chemistry, 21.06.2019 22:30
For the following, determine the type of reaction and then give products.
Answers: 2

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 15:00
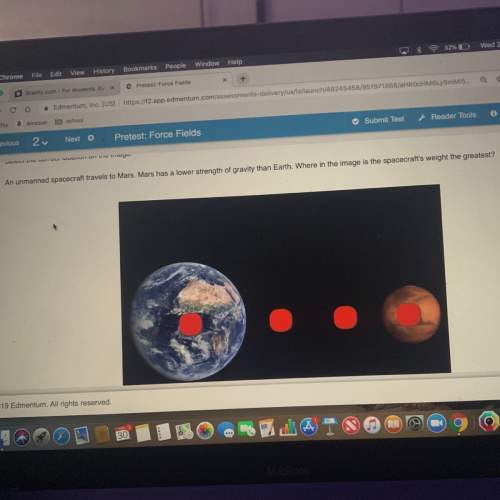
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1
You know the right answer?
A chemistry student needs of carbon tetrachloride for an experiment. By consulting the CRC Handbook...
Questions

Arts, 08.04.2021 17:20


Mathematics, 08.04.2021 17:20

English, 08.04.2021 17:20

History, 08.04.2021 17:20



Business, 08.04.2021 17:20


Mathematics, 08.04.2021 17:20

English, 08.04.2021 17:20

Biology, 08.04.2021 17:20



World Languages, 08.04.2021 17:20

Mathematics, 08.04.2021 17:20