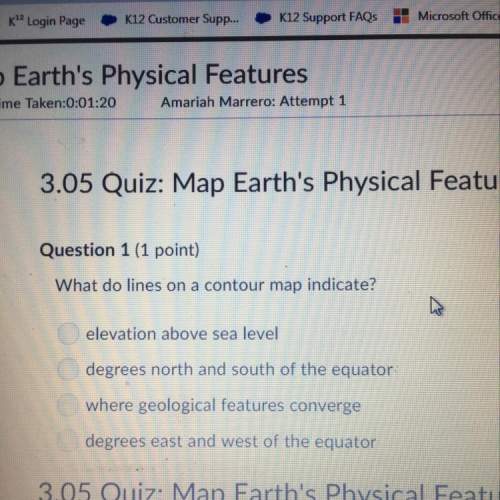
6/10
For the following reaction:
CO + CuO - Cu + CO2
40 g of carbon oxide is reacted wi...

Chemistry, 23.05.2021 01:00 jaylabazemore
6/10
For the following reaction:
CO + CuO - Cu + CO2
40 g of carbon oxide is reacted with 90 g of copper oxide.
Which is the limiting reactant?
(Molar mass of CO = 28 g/mol, molar mass of CuO = 80 g/mol)
a: CO2
b: CuO
c: CO
d: Cu

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 21:30
An atomic nucleus is composed ofa)protons.b)protons and neutrons.c)protons and electrons.d)protons, neutrons, and electrons.
Answers: 1

Chemistry, 23.06.2019 05:30
Scientist think that animals with remarkably similar embryo development probably shared a common ancestor
Answers: 1


Chemistry, 23.06.2019 22:00
2. a sample of table sugar (sucrose, c12h22o11) has a mass of 1.202 g. a. calculate the number of moles of c12h22o11 contained in the sample. show your work. b. calculate the moles of each element in c12h22o11, show your work c. calculate the number of atoms of each type in c12h22o11. show your work
Answers: 2
You know the right answer?
Questions

Social Studies, 18.02.2021 23:00



Chemistry, 18.02.2021 23:00

Mathematics, 18.02.2021 23:00



Spanish, 18.02.2021 23:00

Mathematics, 18.02.2021 23:00






Advanced Placement (AP), 18.02.2021 23:00


Mathematics, 18.02.2021 23:00


Chemistry, 18.02.2021 23:00