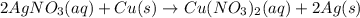Chemistry, 20.05.2021 19:20 deidaraXneji
Study the reaction.
2AgNO3 (aq)+Cu (s)→Cu(NO3)2 (aq)+2Ag (s)
If the oxidation number of copper changes from 0 to +2, is this a redox reaction?
-No; copper gained 2 electrons and was reduced.
-Yes; copper lost 2 electrons and was oxidized.
-Yes, the products have 2 fewer electrons than the reactants.
-No, the products have as many electrons as the reactants.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2


Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3
You know the right answer?
Study the reaction.
2AgNO3 (aq)+Cu (s)→Cu(NO3)2 (aq)+2Ag (s)
If the oxidation number of coppe...
If the oxidation number of coppe...
Questions