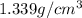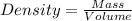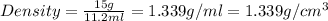Chemistry, 16.10.2019 14:30 jamesgutierrez
Arock weighing 15.0g is placed in a graduated cylinder, displacing the volume from 25.0 ml to 36.2 ml. what is the density of the rock in grams/cubic centimeter?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
Arock weighing 15.0g is placed in a graduated cylinder, displacing the volume from 25.0 ml to 36.2 m...
Questions



Mathematics, 05.07.2019 11:20




Mathematics, 05.07.2019 11:30

History, 05.07.2019 11:30

Chemistry, 05.07.2019 11:30

Health, 05.07.2019 11:30

Social Studies, 05.07.2019 11:30

Mathematics, 05.07.2019 11:30

Social Studies, 05.07.2019 11:30




Business, 05.07.2019 11:30