Chemistry, 12.04.2021 22:00 macylen3900
(1st Screenshot)
(a) Using the information in the table above, determine the value of ΔG° at 298K for the process represented by the equation H2O(l)⇄H2O(g).
(b) Considering your answer to part (a), indicate whether the process is thermodynamically favorable at 298K. Justify your answer.
(c) Considering your answer to part (b), explain why H2O(l) has a measurable equilibrium vapor pressure at 298K.
(2nd Screenshot)
Water vapor can be produced in two different processes, as represented below.
(d) In terms of concepts of entropy and the particle-level structure of the different phases of water, explain why the change in entropy, ΔS, is greater for process 1 than for process 2.
Please help as soon as possible


Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
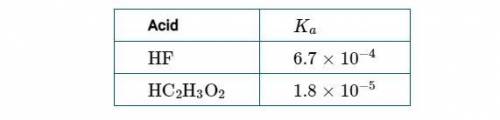
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1

You know the right answer?
(1st Screenshot)
(a) Using the information in the table above, determine the value of ΔG° at 298K f...
Questions


Mathematics, 28.01.2020 12:31

History, 28.01.2020 12:31

Social Studies, 28.01.2020 12:31


History, 28.01.2020 12:31

History, 28.01.2020 12:31







Chemistry, 28.01.2020 12:31

English, 28.01.2020 12:31





Biology, 28.01.2020 12:31