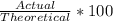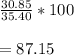Chemistry, 29.03.2021 17:10 varonadestiny1109
What is the percent yield of LiCl if I produced 30.85g LiCl and my theoretical yield was calculated to be 35.40g LiCl?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
You know the right answer?
What is the percent yield of LiCl if I produced 30.85g LiCl and my theoretical yield was calculated...
Questions


Computers and Technology, 27.09.2019 14:10





Mathematics, 27.09.2019 14:10

Biology, 27.09.2019 14:10

Mathematics, 27.09.2019 14:10

Physics, 27.09.2019 14:10


Social Studies, 27.09.2019 14:10





Social Studies, 27.09.2019 14:10


Chemistry, 27.09.2019 14:10

Social Studies, 27.09.2019 14:10