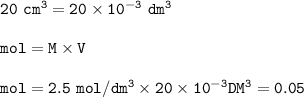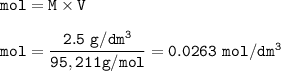Chemistry, 27.12.2020 03:00 sunarafet3814
1. Magnesium chloride solution reacts with silver nitrate solution to form magnesium nitrate
solution and silver chloride.
Equation: MgCl2 (s) + 2 AgNO3 (aq) → Mg(NO3)2 (aq) + 2 AgCl (s)
Find the mass of silver chloride formed if
(a) 20 cm of 2.5 mol/dm^3 of magnesium chloride solution is used.
(6) 20 cm of 2.5 g/dm^3 of magnesium chloride solution is used.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 14:30
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1

Chemistry, 23.06.2019 01:30
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1

Chemistry, 23.06.2019 13:20
Which nuclide is most likely to be radioactive and synthetic 24/12 mg237/93mg195/78mg230/84mg
Answers: 1
You know the right answer?
1. Magnesium chloride solution reacts with silver nitrate solution to form magnesium nitrate
soluti...
Questions


English, 13.01.2020 09:31


Computers and Technology, 13.01.2020 09:31

Mathematics, 13.01.2020 09:31




Mathematics, 13.01.2020 09:31





Mathematics, 13.01.2020 09:31

Arts, 13.01.2020 09:31

Geography, 13.01.2020 09:31

Mathematics, 13.01.2020 09:31