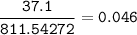Chemistry, 21.11.2020 01:50 alexismoore15567
Consider the reaction of lead(II) nitrate reacting with sodium phosphate to create lead(II) phosphate and sodium nitrate. If 37.1 g of lead(II) phosphate was created in the reaction, how much (mass) sodium phosphate was originally needed to produce this amount of product

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Asolution is made by dissolving 25.5 grams of glucose (c6h12o6) in 398 grams of water. what is the freezing point depression of the solvent if the freezing point constant is -1.86 °c/m? show all of the work needed to solve this problem.
Answers: 1

Chemistry, 22.06.2019 03:30
What is the number of moles of chemical units represented by 9.03x10^24? and how do i show work? (dumb it down )
Answers: 1

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1
You know the right answer?
Consider the reaction of lead(II) nitrate reacting with sodium phosphate to create lead(II) phosphat...
Questions



Mathematics, 29.01.2020 10:59


English, 29.01.2020 10:59


Mathematics, 29.01.2020 10:59

Social Studies, 29.01.2020 10:59

Mathematics, 29.01.2020 10:59


Biology, 29.01.2020 10:59

Mathematics, 29.01.2020 10:59

Arts, 29.01.2020 10:59


Mathematics, 29.01.2020 11:00





History, 29.01.2020 11:00