
Chemistry, 20.11.2020 14:00 ymklay1566
What mass of sucrose, C12H22O11, should you dissolve in 100.0 g of water to obtain a solution in which the mole fraction of C12H22O11 is 0.124?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3


Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

Chemistry, 23.06.2019 07:00
Why do the strengths of london (dispersion) forces generally increase with increasing molecular size? choose one: a. heavier atoms have stronger attractions for each other than lighter atoms. b. dispersion forces are all equal in magnitude; there is no size dependence. c. dispersion forces arise from the attraction between the nuclei of atoms, and larger molecules have larger nuclei. d. dispersion forces arise from dipoles caused by the electron distribution being distorted. larger molecules have more electrons and, therefore, more distortions and a bigger force. e. dispersion forces depend on distance. larger molecules are farther apart and so the forces are smaller.
Answers: 2
You know the right answer?
What mass of sucrose, C12H22O11, should you dissolve in 100.0 g of water to obtain a solution in whi...
Questions





Mathematics, 06.05.2020 05:37


Mathematics, 06.05.2020 05:37


Mathematics, 06.05.2020 05:37


English, 06.05.2020 05:37


History, 06.05.2020 05:37

English, 06.05.2020 05:37


Computers and Technology, 06.05.2020 05:37




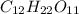 is 342.3 g/mol
is 342.3 g/mol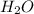 is 18g/mol
is 18g/mol


