Chemistry, 27.10.2020 21:30 mbonham481
A sample of Ar gas has a pressure of 1.20 atm at 125°C and a volume of 1.00 L. What would the temperature be (°C) with a pressure of 1.80 atm and constant volume?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1


Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2
You know the right answer?
A sample of Ar gas has a pressure of 1.20 atm at 125°C and a volume of 1.00 L. What would the temper...
Questions

History, 07.04.2020 19:30






Advanced Placement (AP), 07.04.2020 19:30




Mathematics, 07.04.2020 19:30

Biology, 07.04.2020 19:30

History, 07.04.2020 19:30

Mathematics, 07.04.2020 19:30






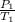 =
= 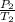
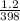 =
=