
Chemistry, 20.10.2020 01:01 aaronjin4443
PLz HELP ASAP! Mass of stoppered test tube plus metal 95 g
Mass of test tube and stopper 31.3g
Mass of calorimeter 2.5 g
Mass of calorimeter and water 46.1 g
Mass of water (subtraction) _ g
Mass of metal (subtraction) _ g
Initial temperature of water in calorimeter 24.0°C
Initial temperature of metal (assume 100°C
unless directed to do otherwise) 98.4°C
Equilibrium temperature of metal and water
in calorimeter 29.0°C
Δ 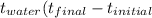 _ °C
_ °C
Δ 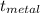 _ °C
_ °C
_ J
Specific heat of the metal _ J/g°C
Approximate molar mass of metal _ g/mol
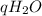

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

You know the right answer?
PLz HELP ASAP! Mass of stoppered test tube plus metal 95 g
Mass of test tube and stopper 31.3g
Questions

English, 26.11.2019 14:31


English, 26.11.2019 14:31

Social Studies, 26.11.2019 14:31


Social Studies, 26.11.2019 14:31


History, 26.11.2019 14:31




Biology, 26.11.2019 14:31


History, 26.11.2019 14:31


Geography, 26.11.2019 14:31



Mathematics, 26.11.2019 14:31

History, 26.11.2019 14:31



