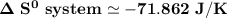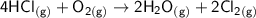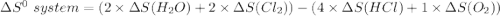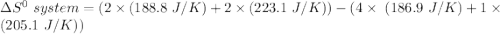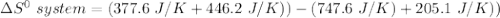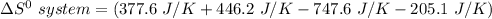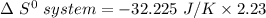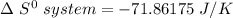Chemistry, 26.08.2020 15:01 rayvingrant16
Consider the reaction: 4HCl(g) + O2(g)2H2O(g) + 2Cl2(g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 2.23 moles of HCl(g) react at standard conditions. S°system = J/K

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 14:40
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1
You know the right answer?
Consider the reaction: 4HCl(g) + O2(g)2H2O(g) + 2Cl2(g) Using standard absolute entropies at 298K, c...
Questions


Mathematics, 01.10.2019 17:30

History, 01.10.2019 17:30


Geography, 01.10.2019 17:30






Mathematics, 01.10.2019 17:30



Mathematics, 01.10.2019 17:30


Social Studies, 01.10.2019 17:30

English, 01.10.2019 17:30

Mathematics, 01.10.2019 17:30

Biology, 01.10.2019 17:30