
Chemistry, 29.05.2020 06:02 angelZ3947
The first two steps in the industrial synthesis of nitric acid produce nitrogen dioxide from ammonia: The net reaction is: Write an equation that gives the overall equilibrium constant in terms of the equilibrium constants and . If you need to include any physical constants, be sure you use their standard symbols, which you'll find in the ALEKS Calculator.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 20:40
What effect would average population growth have on land usage? a. urban use of land would rise to more than 30 percent of available land. b. industrial use of land would rise to more than 30 percent of available land. c. the percentage of available land used as cropland would stay the same. d. cropland would fall to about 10 percent of available land.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the mass of naoh that would have to be added to 500 ml of a solution of 0.20 m acetic acid in order to achieve a ph of 5.0?
Answers: 1

Chemistry, 23.06.2019 00:30
Which radioisotope is used to date fossils? a. oxygen-16 b. carbon-14 c. uranium-238 d. carbon-12
Answers: 2
You know the right answer?
The first two steps in the industrial synthesis of nitric acid produce nitrogen dioxide from ammonia...
Questions




Business, 23.09.2019 07:10

Social Studies, 23.09.2019 07:10



Social Studies, 23.09.2019 07:10


History, 23.09.2019 07:10

Mathematics, 23.09.2019 07:10


English, 23.09.2019 07:10


Mathematics, 23.09.2019 07:10

English, 23.09.2019 07:10

English, 23.09.2019 07:10

Biology, 23.09.2019 07:10

History, 23.09.2019 07:10

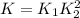
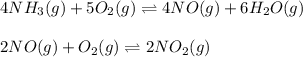
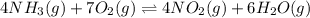
![K_1=\frac{[NO]^4[H_2O]^6}{[NH_3]^4[O_2]^5}\\ \\K_2=\frac{[NO_2]^2}{[NO]^2[O_2]}](/tpl/images/0669/9771/004ed.png)
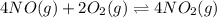
![K_2^{new}=\frac{[NO_2]^4}{[NO]^4[O_2]^2}](/tpl/images/0669/9771/b0ba1.png)
![K_1*K_2^2=\frac{[NO]^4[H_2O]^6}{[NH_3]^4[O_2]^5}*\frac{[NO_2]^4}{[NO]^4[O_2]^2}=\frac{[H_2O]^6[NO_2]^4}{[NH_3]^4[O_2]^7}](/tpl/images/0669/9771/5162f.png)
![K=\frac{[H_2O]^6[NO_2]^4}{[NH_3]^4[O_2]^7}](/tpl/images/0669/9771/dc773.png)


