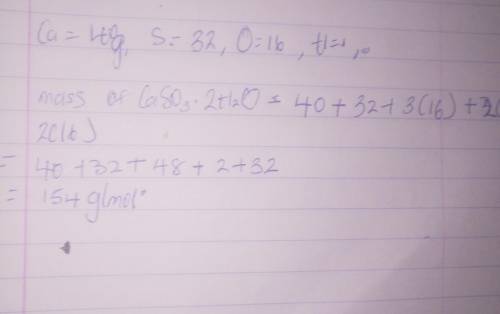Answers: 3


Another question on Chemistry


Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
What is the mass of CaSO3 •2H2O...
Questions

English, 29.09.2019 20:00

English, 29.09.2019 20:00



Chemistry, 29.09.2019 20:00

Mathematics, 29.09.2019 20:00

Biology, 29.09.2019 20:00


Mathematics, 29.09.2019 20:00

History, 29.09.2019 20:00

Mathematics, 29.09.2019 20:00






Social Studies, 29.09.2019 20:00

Social Studies, 29.09.2019 20:00