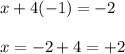Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1


Chemistry, 22.06.2019 06:30
The minerals found in bones are deposited by living cells called
Answers: 1

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2
You know the right answer?
What is the oxidation number for zn(oh)4 2-?...
Questions

Chemistry, 08.10.2020 04:01


Mathematics, 08.10.2020 04:01




Biology, 08.10.2020 04:01

Mathematics, 08.10.2020 04:01

Mathematics, 08.10.2020 04:01

Mathematics, 08.10.2020 04:01



Mathematics, 08.10.2020 04:01




Mathematics, 08.10.2020 04:01

Mathematics, 08.10.2020 04:01

Mathematics, 08.10.2020 04:01

![[Zn(OH)_{4}]^{2-}](/tpl/images/0482/1498/eb61f.png)