
Chemistry, 25.04.2020 03:46 breannaking9734
Sodium chloride, NaCl forms in this reaction between sodium and chlorine. 2Na(s) + Cl2(g) → 2NaCl(s) How many moles of NaCl result from the complete reaction of 3.9 mol of Cl2? Assume that there is more than enough Na.

Answers: 3


Another question on Chemistry

Chemistry, 23.06.2019 02:30
Which of the four hypothetical substances you investigated would be most harmful to living organisms? 50 points!
Answers: 2

Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1

Chemistry, 23.06.2019 09:20
Four statements about the development of the atomic model are shown below. a: electrons have wavelike properties. b: atoms have small, negatively charged particles. c. the center of an atom is a small, dense nucleus. d: atoms are hard, indivisible spheres. which order of statements represents the historical development of the atomic model? c-d-a-b c-d-b-a d— в-а — с d-b-c-a
Answers: 1

Chemistry, 23.06.2019 10:00
Which of the following reasons best explains why a scientist would want to replicate gregor mendel's pea plant experiment? a. to discover new aspects of the natural world b. to test the predictions of current theories c. to explain recently observed phenomena d. to test the conclusions of prior investigations
Answers: 1
You know the right answer?
Sodium chloride, NaCl forms in this reaction between sodium and chlorine. 2Na(s) + Cl2(g) → 2NaCl(s)...
Questions

Mathematics, 11.07.2019 01:00









Mathematics, 11.07.2019 01:00

Mathematics, 11.07.2019 01:00


Advanced Placement (AP), 11.07.2019 01:00

Computers and Technology, 11.07.2019 01:00

Mathematics, 11.07.2019 01:00



Computers and Technology, 11.07.2019 01:00


Mathematics, 11.07.2019 01:00

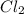
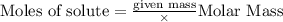
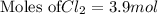
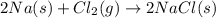
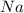 is the excess reagent,
is the excess reagent, 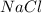
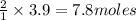 of
of 

