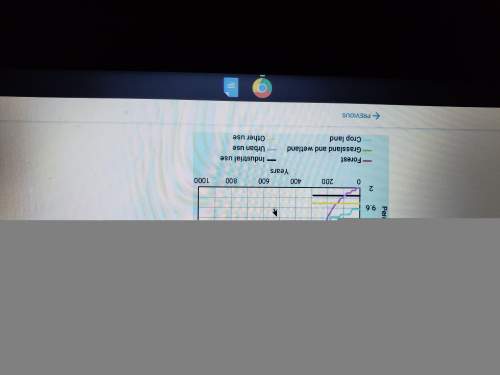
2. Write the equilibrium expression for HF + H2O → F- + H3O+
(i) Find [H+] of a 0.056 M...

2. Write the equilibrium expression for HF + H2O → F- + H3O+
(i) Find [H+] of a 0.056 M hydrofluoric acid solution. Ka = 1.45 x 10-7
(ii) Then calculate the pH
3. Write the equilibrium expression for NH3 + H2O → NH4+ + OH-
4. Write the equilibrium expression for H2S + H2O → H3O+ + HS- and
(i) Calculate the [H3O+] in a 0.45 M solution of hydrogen sulphide (H2S). Ka = 2.05 x 10-6,
(ii)Then calculate the pH of H2S?
5. Write the equilibrium expression for NH3 + H2O g NH4+ + OH-
(i) Calculate the [OH-] in a 0.60 M solution of ammonia (NH3) Kb = 6.83 X 10-4,
(ii)Then calculate the pHof NH3 (Base)
6. The pH in a 0.25 M solution of the acid HBrO is 4.65 . Using this, Write the equilibrium expression of
HBrO and calculate the value of Ka for the acid HBrO

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
You know the right answer?
Questions

Computers and Technology, 02.07.2019 17:30




Social Studies, 02.07.2019 17:30



Biology, 02.07.2019 17:30

Mathematics, 02.07.2019 17:30




English, 02.07.2019 17:30





History, 02.07.2019 17:30

Social Studies, 02.07.2019 17:30

Biology, 02.07.2019 17:30