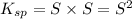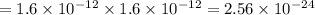Chemistry, 01.04.2020 22:35 glocurlsprinces
The molar solubility of zns is 1.6 Ã 10-12 m in pure water. Calculate the ksp for zns.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
A6.10 m nacl can be made by adding [x]g of nacl to a container and making the volume of water up to the 1.00 l line
Answers: 1

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 20:40
What effect would average population growth have on land usage? a. urban use of land would rise to more than 30 percent of available land. b. industrial use of land would rise to more than 30 percent of available land. c. the percentage of available land used as cropland would stay the same. d. cropland would fall to about 10 percent of available land.
Answers: 1
You know the right answer?
The molar solubility of zns is 1.6 Ã 10-12 m in pure water. Calculate the ksp for zns....
Questions


History, 26.03.2021 08:20


Arts, 26.03.2021 08:20

Mathematics, 26.03.2021 08:20

Mathematics, 26.03.2021 08:20

Mathematics, 26.03.2021 08:20



Mathematics, 26.03.2021 08:20

Mathematics, 26.03.2021 08:20








Mathematics, 26.03.2021 08:20

World Languages, 26.03.2021 08:20

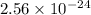 .
.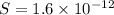
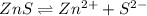
![K_{sp}=[Zn^{2+}]\times [S^{2-}]](/tpl/images/0576/5742/dbee8.png)