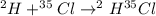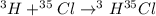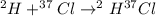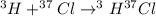Chemistry, 17.03.2020 05:22 TheViperMlg23676
Hydrogen and chlorine atoms react to form a simple diatomic molecule in a 1:1 ration, that is HCl. The percent isotopic abundances of the chlorine isotopes 35Cl and 37Cl are 75.77% and 24.23%, respectively. The percent isotopic abundances of the hydrogen isotopes are 2H and 3H are 0.015% and 0.001%. (a) How many different HCl molecules are possible, and what are their mass numbers

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:10
Which of these will change if the air in aclosed bottle is heated? abcdthe mass of the airthe composition of the airthe air pressure in the bottlethe number of air molecules in the bottle
Answers: 3

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

You know the right answer?
Hydrogen and chlorine atoms react to form a simple diatomic molecule in a 1:1 ration, that is HCl. T...
Questions




Computers and Technology, 23.01.2022 03:20





Biology, 23.01.2022 03:20



Business, 23.01.2022 03:20

Business, 23.01.2022 03:20







Biology, 23.01.2022 03:20

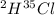 = 37 amuMass of
= 37 amuMass of 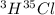 = 38 amuMass of
= 38 amuMass of 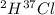 = 39 amuMass of
= 39 amuMass of 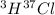 = 40 amu
= 40 amu