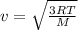Chemistry, 11.03.2020 23:08 dgonzale1857
(CO2) (O2) (He) (N2) (CH4) (Imagine these are balloons with the specified gas inside)
Above are five identical balloons, each filled to the same volume at 25°C and 1.0 atm
Which balloon contains the molecules with the greatest average speed? Which has the Lowest average speed?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2

Chemistry, 22.06.2019 12:30
The missing component to the table to the right or indicated with orange letters complete the table by filling in the corresponding numbers or symbols
Answers: 3

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1
You know the right answer?
(CO2) (O2) (He) (N2) (CH4) (Imagine these are balloons with the specified gas inside)
Ab...
Ab...
Questions

Chemistry, 09.12.2020 22:30


Business, 09.12.2020 22:30

History, 09.12.2020 22:30

English, 09.12.2020 22:30


English, 09.12.2020 22:30


Physics, 09.12.2020 22:30



Biology, 09.12.2020 22:30

Advanced Placement (AP), 09.12.2020 22:30

Mathematics, 09.12.2020 22:30

Mathematics, 09.12.2020 22:30


History, 09.12.2020 22:30

Business, 09.12.2020 22:30