
Chemistry, 05.03.2020 05:16 calmicaela12s
Consider the unbalanced equation for the combustion of hexane: C6H14 (g) O2 (g) --> CO2 (g) H2O (g) Balance the equation and determine how many moles of O2 are required to react completely with 7.2 moles of C6H14.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 23.06.2019 00:20
How many lone pairs of electrons are on the central atom of no3- and what is the molecular shape? one, trigonal planar zero, trigonal pyramidal zero, trigonal planar one, tetrahedral one, trigonal pyramidal
Answers: 1
You know the right answer?
Consider the unbalanced equation for the combustion of hexane: C6H14 (g) O2 (g) --> CO2 (g) H2O (...
Questions

Mathematics, 12.03.2021 04:50


Mathematics, 12.03.2021 04:50



Mathematics, 12.03.2021 04:50

Mathematics, 12.03.2021 04:50



Social Studies, 12.03.2021 04:50



Mathematics, 12.03.2021 04:50



English, 12.03.2021 05:00


Mathematics, 12.03.2021 05:00


History, 12.03.2021 05:00

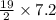 .
.

