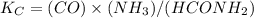Chemistry, 25.02.2020 06:13 BIKRAMlawati5544
Onsider the reaction, which takes place at a certain elevated temperature CO(g)+NH3(g)⇌HCONH2(g), Kc=0.860 If a reaction vessel initially contains only CO and NH3 at concentrations of 1.00 M and 2.00 M, respectively, what will the concentration of HCONH2 be at equilibrium?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3


Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

Chemistry, 23.06.2019 02:00
As light moves from one material into the next, which of the following affects how much the light waves will refract, or bend? angle at which the ray strikes the medium color of the material density of the material temperature of the light wave
Answers: 2
You know the right answer?
Onsider the reaction, which takes place at a certain elevated temperature CO(g)+NH3(g)⇌HCONH2(g), Kc...
Questions

Mathematics, 04.12.2020 05:00


Mathematics, 04.12.2020 05:00


Chemistry, 04.12.2020 05:00






Mathematics, 04.12.2020 05:00





Mathematics, 04.12.2020 05:00