
Give the charge and full ground-state electron configuration of the monatomic ion most likely to be formed by each element:. a. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 b. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p2 c. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p4 d. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p8e. None of these

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 16:00
Water is called the universal solvent because more substances dissolve in water than in any other chemical. this has to do with the polarity of each water molecule. the hydrogen side of each water (h2o) molecule carries a slight positive electric charge, while the oxygen side carries a slight negative electric charge.
Answers: 3

Chemistry, 23.06.2019 16:00
Henry moseley used x-ray experiments to determine the atomic number of elements. how did his discovery contribute to the development of the periodic table? a.it confirmed that elements should be arranged in strict order of increasing atomic mass. b.it led to elements with similar atomic numbers being grouped together. c.it allowed the elements to be placed in strict order of increasing atomic number. d.it showed that the way mendeleev grouped elements together was completely wrong.
Answers: 1

Chemistry, 23.06.2019 17:00
Identify the missing coefficient in the balanced equation and classify the type of reaction. mg(oh)2 + h2so4 ⟶ mgso4 + 1; combustion 1; neutralization 2; combustion 2; neutralization
Answers: 1
You know the right answer?
Give the charge and full ground-state electron configuration of the monatomic ion most likely to be...
Questions




Mathematics, 21.03.2020 04:52

Mathematics, 21.03.2020 04:52

Mathematics, 21.03.2020 04:52

English, 21.03.2020 04:52



Spanish, 21.03.2020 04:52




Biology, 21.03.2020 04:53



Mathematics, 21.03.2020 04:53



Mathematics, 21.03.2020 04:54

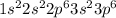
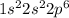
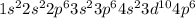
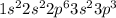
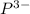 ion
ion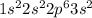
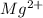 ion
ion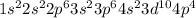
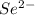 ion
ion

