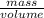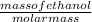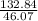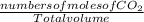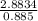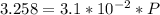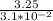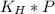Chemistry, 19.02.2020 01:04 hilario4785
Fermentation of 826 mL grape juice (density is 1.0 ) is allowed to take place in a bottle with a total volume of 885 mL until 19% by volume is ethanol (). Assuming that obeys Henry’s law, calculate the partial pressure of in the gas phase and the solubility of in the wine at 25°C. The Henry’s law constant for is mol/L ⋅ atm at 25°C with Henry’s law in the form , where is the concentration of the gas in mol/L. (The density of ethanol is 0.79 .)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Y=‐1x + 7 if y has a value of ‐24 what is the value of x?
Answers: 1

Chemistry, 23.06.2019 01:00
Which statement characterizes synthetic polymers? a. they come from animals and plants. b. they are found in nature. c. they are made in a lab. d. they are components of starch.
Answers: 1

Chemistry, 23.06.2019 10:00
Which number should be placed before f2 on the reactants side equation to make equation balanced? xe + > xef4
Answers: 1

Chemistry, 23.06.2019 17:20
What is the relationship between ka and kb with kw? the sum of ka and kb equals the auto-dissociation constant for water. the product of ka and kb equals the auto-dissociation constant for water. the quotient of ka and kb equals the auto-dissociation constant for water. the difference of ka and kb equals the auto-dissociation constant for water.
Answers: 3
You know the right answer?
Fermentation of 826 mL grape juice (density is 1.0 ) is allowed to take place in a bottle with a tot...
Questions



Mathematics, 12.04.2021 23:30

Mathematics, 12.04.2021 23:30

Mathematics, 12.04.2021 23:30

Computers and Technology, 12.04.2021 23:30


Mathematics, 12.04.2021 23:30

Mathematics, 12.04.2021 23:30




Computers and Technology, 12.04.2021 23:30


English, 12.04.2021 23:30

Physics, 12.04.2021 23:30