
Chemistry, 13.02.2020 05:55 jasonoliva13
Which of the following sets are isoelectronic (i. e., have the same number of electrons)? i. Br-, Kr, Sr2+ ii. C, N-, O2- iii. Mg2+, Ca2+, Sr2+ iv. O2-, O, O2+ v. Ag+, Cd2+, Pd

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1



Chemistry, 23.06.2019 13:30
Determine the rate law, including the values of the orders and rate law constant, for the following reaction using the experimental data provided. a + b yields products trial [a] [b] rate 1 0.30 m 0.25 m 1.2 × 10-2 m/min 2 0.30 m 0.50 m 4.8 × 10-2 m/min 3 0.60 m 0.50 m 9.6 × 10-2 m/min
Answers: 1
You know the right answer?
Which of the following sets are isoelectronic (i. e., have the same number of electrons)? i. Br-, Kr...
Questions

Biology, 28.11.2019 18:31


World Languages, 28.11.2019 18:31


Computers and Technology, 28.11.2019 18:31

Advanced Placement (AP), 28.11.2019 18:31



Social Studies, 28.11.2019 18:31







History, 28.11.2019 18:31




Social Studies, 28.11.2019 18:31

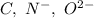
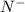 has 6 electrons.
has 6 electrons.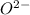 has 6 electrons.
has 6 electrons.

