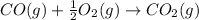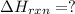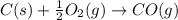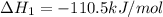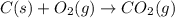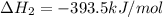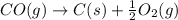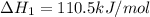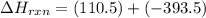CO(g) + 12 O2(g) → CO2(g)The combustion of carbon monoxide is represented by the equation above.(a) Determine the value of the standard enthalpy change, ∆HrxnD , for the combustion of CO(g) at 298 Kusing the following information. C(s) + 12 O2(g) → CO(g) ∆H298D = − 110.5 kJ mol−1 C(s) + O2(g) → CO2(g) ∆H298D = − 393.5 kJ mol−1

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
The isotonic saline solution described in part a is connected to an unknown solution via a semipermeable membrane, the unknown solution level drops. based on this information, what can be said about these two solutions?
Answers: 1

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1

Chemistry, 23.06.2019 00:50
50 points! need answer asap. what type of organic compound contains the following functional group? (2 points)
Answers: 3
You know the right answer?
CO(g) + 12 O2(g) → CO2(g)The combustion of carbon monoxide is represented by the equation above.(a)...
Questions

Mathematics, 09.10.2019 16:20

History, 09.10.2019 16:20

World Languages, 09.10.2019 16:20


Mathematics, 09.10.2019 16:20

SAT, 09.10.2019 16:20



Chemistry, 09.10.2019 16:20











 will be,
will be,