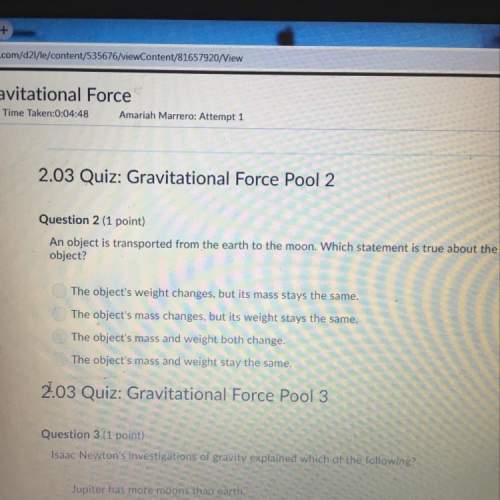
What is the term for the molecular mass of any substance expressed in grams?
a. molar ma...

Chemistry, 15.11.2019 22:31 Jamilia561
What is the term for the molecular mass of any substance expressed in grams?
a. molar mass
b. molar volume
c. molar density
d. mole

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
The most efficient way to establish the best possible economizer position is to measure
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
Questions


Mathematics, 20.08.2019 17:30



Social Studies, 20.08.2019 17:30

Mathematics, 20.08.2019 17:30

Mathematics, 20.08.2019 17:30


History, 20.08.2019 17:30





English, 20.08.2019 17:30


Social Studies, 20.08.2019 17:30

Mathematics, 20.08.2019 17:30

Mathematics, 20.08.2019 17:30


Mathematics, 20.08.2019 17:30