
Chemistry, 01.11.2019 02:31 scarletmartinez
According to the arrhenius concept, an acid is a substance that a. is capable of donating one or more h⁺b. tastes bitter c. reacts with the solvent to form the cation formed by autoionization of that solvent d. causes an increase in the concentration of h⁺ in aqueous solutions e. can accept a pair of electrons to form a coordinate covalent bond

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
You know the right answer?
According to the arrhenius concept, an acid is a substance that a. is capable of donating one or mo...
Questions

Mathematics, 25.01.2021 07:20


Mathematics, 25.01.2021 07:20


Mathematics, 25.01.2021 07:20

Mathematics, 25.01.2021 07:20

Mathematics, 25.01.2021 07:20


History, 25.01.2021 07:30







Chemistry, 25.01.2021 07:30

Biology, 25.01.2021 07:30


Spanish, 25.01.2021 07:30

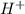 .
.

