
Chemistry, 16.09.2019 21:30 he0gaubong
Which of the following statements about acids and bases are true? acetic acid is a strong acid. a strong acid has a weak conjugate base. a brønsted base is a proton acceptor. bases need not contain –oh directly; instead, they can increase the concentration of –oh by deprotonating water.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2


Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 15:30
The identities of substances are the same before and after which type of change
Answers: 1
You know the right answer?
Which of the following statements about acids and bases are true? acetic acid is a strong acid. a st...
Questions



Mathematics, 07.01.2021 01:00

Biology, 07.01.2021 01:00

English, 07.01.2021 01:00

Mathematics, 07.01.2021 01:00

Mathematics, 07.01.2021 01:00

History, 07.01.2021 01:00

Arts, 07.01.2021 01:00



History, 07.01.2021 01:00

History, 07.01.2021 01:00

Health, 07.01.2021 01:00






History, 07.01.2021 01:00

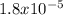 , pKa will be
, pKa will be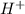 and
and 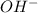 directly, they can increase their concentration by deprotonating water.
directly, they can increase their concentration by deprotonating water. 

