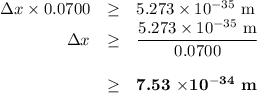Chemistry, 14.09.2019 11:30 cjstablet04
Consider an electron with a mass of 9.11 x 1051 kg and a 100.0 g tennis ball that are both moving with a velocity of 70.0 m s1. (a) calculate the momentum of the electron (p mv). (b) calculate the momentum of the tennis ball (c) what is the uncertainty in the position of the electron (ax) if the uncertainty in its momentum (ap) is equal to 1.0 % of p for the electron? (d) what is the uncertainty in the position of the tennis ball (ax) if the uncertainty in momentum (ap) is equal to 0.1% of p for the tennis ball? (e) comment on how the uncertainty in position (ax) compares to the overall size in each case

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1

Chemistry, 23.06.2019 06:10
How can liquids be seperated by density a the liquids are absorbed onto a paper b the liquids are turned into seperate vapors c the liquids are collected as they evaporate d the liquids are allowed to seperate into layers
Answers: 1

Chemistry, 23.06.2019 06:30
What happens to the glucose molecule during the process of cellular respiration? (5 points) select one: a. it gets broken down. b. it forms oxygen. c. it builds muscles. d. it uses up energy.
Answers: 3
You know the right answer?
Consider an electron with a mass of 9.11 x 1051 kg and a 100.0 g tennis ball that are both moving wi...
Questions