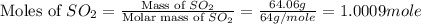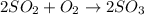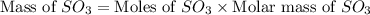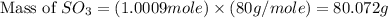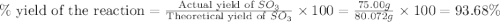In the manufacturing process of sulfuric acid, sulfur dioxide is reacted with oxygen to produce sulfur trioxide. using the equation, 2so2 (g) + o2 imported asset 2so3 (g), if 64.06g of sulfur dioxide is given an opportunity to react with an excess of oxygen to produce 75.00 g of sulfur trioxide, what is the percent yield of this reaction? 46.83% 60.25% 75.55% 93.68%

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1


Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
You know the right answer?
In the manufacturing process of sulfuric acid, sulfur dioxide is reacted with oxygen to produce sulf...
Questions

Biology, 12.07.2019 02:00



Mathematics, 12.07.2019 02:00

History, 12.07.2019 02:00

History, 12.07.2019 02:00




Health, 12.07.2019 02:00

Computers and Technology, 12.07.2019 02:00

Geography, 12.07.2019 02:00


Geography, 12.07.2019 02:00

Social Studies, 12.07.2019 02:00


Geography, 12.07.2019 02:00



Biology, 12.07.2019 02:00

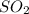 = 64.06 g
= 64.06 g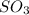 = 80 g/mole
= 80 g/mole