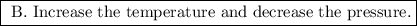** answer fast i will mark you brainliest and give 20 points * * this is a chemistry question so only answer if you are good at chemistry *
a certain reaction is endothermic in the forward direction and has a greater number of molecules on the product side. which set of conditions should be used to maximize the yield of the products?
a. decrease the temperature and decrease the pressure.
b. increase the temperature and decrease the pressure.
c. decrease the temperature and increase the pressure.
d. increase the temperature and increase the pressure.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
What postulate of the kinetic molecular theory best explains why gases have high fluidity? because collisions between gas particles are elastic, there is no loss of energy as particles flow past each other. because gases consist of large numbers of tiny particles, they spread out and do not come in contact with each other. because the attractive forces between gas particles are negligible, gas particles can glide easily past one another. because the average kinetic energy of gas particles increases as temperature increases, gas particles behave more like a liquid. question 6 compare the compressibility of gases and liquids. support your answer by describing the arrangement of particles in gases and liquids.
Answers: 1

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3


Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 1
You know the right answer?
** answer fast i will mark you brainliest and give 20 points * * this is a chemistry question so on...
Questions

Computers and Technology, 07.06.2020 02:59

Mathematics, 07.06.2020 02:59

Mathematics, 07.06.2020 02:59



History, 07.06.2020 02:59

History, 07.06.2020 02:59

Mathematics, 07.06.2020 02:59






Chemistry, 07.06.2020 02:59

History, 07.06.2020 02:59

Biology, 07.06.2020 03:00


Mathematics, 07.06.2020 03:00

Chemistry, 07.06.2020 03:00

Biology, 07.06.2020 03:00