Use the drop-down menus to identify the electromagnetic waves.
1.) infrared
2.) gamm...

Chemistry, 21.01.2020 21:31 jakhunter354
Use the drop-down menus to identify the electromagnetic waves.
1.) infrared
2.) gamma rays
3.) radio waves
4.) visible light
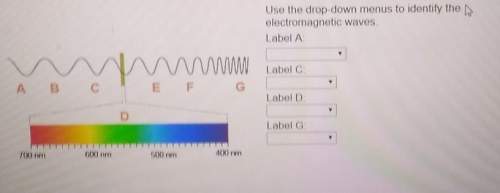

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
Questions


Arts, 29.07.2020 07:01




Mathematics, 29.07.2020 07:01





English, 29.07.2020 07:01

Social Studies, 29.07.2020 07:01

Mathematics, 29.07.2020 07:01

Mathematics, 29.07.2020 07:01


Mathematics, 29.07.2020 07:01

Mathematics, 29.07.2020 07:01



Mathematics, 29.07.2020 07:01



