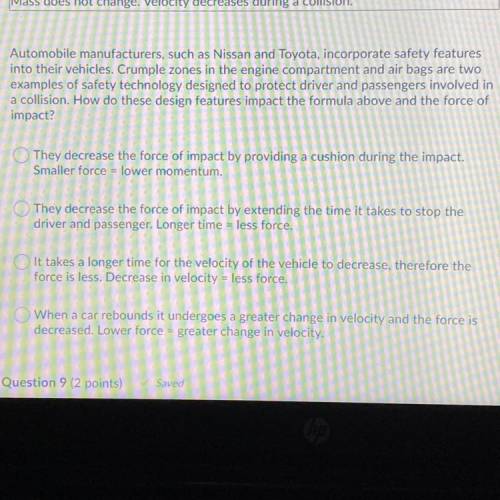
Will give correct answer brainliest
...

Answers: 3


Another question on Physics



Physics, 22.06.2019 05:30
If gases like carbon dioxide and methane make up less than 1% of the total atmosphere, why is it important for scientists to monitor changes in percentages of these gases?
Answers: 1

Physics, 22.06.2019 11:00
1.)the isotope cobalt-60 has a nuclear mass of 59.933820 u calculate the mass defect of cobalt-60 using the following information. mass of proton: 1.007825 u mass of neutron: 1.008665 u 1 u = 931.5 mev 2.)the isotope cobalt-60 has a nuclear mass of 59.933820 u calculate the binding energy of cobalt-60 using the following information. mass of proton: 1.007825 u mass of neutron: 1.008665 u 1 u = 931.5 mev 3.)the isotope cobalt-60 has a nuclear mass of 59.933820 u calculate the binding energy per nucleon of cobalt-60 using the following information. mass of proton: 1.007825 u mass of neutron: 1.008665 u 1 u = 931.5 mev
Answers: 3
You know the right answer?
Questions

History, 23.07.2019 04:30

Chemistry, 23.07.2019 04:30

History, 23.07.2019 04:30

History, 23.07.2019 04:30


Social Studies, 23.07.2019 04:30


History, 23.07.2019 04:30

Social Studies, 23.07.2019 04:30








Social Studies, 23.07.2019 04:40