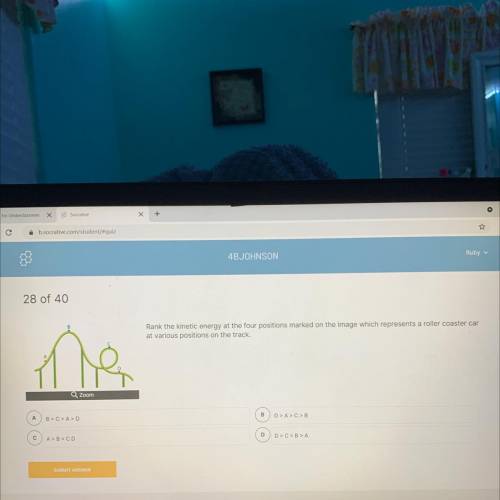
Helppp please. NO LINKS. HELP HELP HELP
...

Answers: 3


Another question on Physics



Physics, 22.06.2019 19:30
A47.2 g block of copper whose temperature is 480 k is placed in an insulating box with a 91.8 g block of lead whose temperature is 200 k. (a) what is the equilibrium temperature of the two-block system? (b) what is the change in the internal energy of the two-block system between the initial state and the equilibrium state? (c) what is the change in the entropy of the two-block system? the heat capacities of copper and lead are 386 j/kg·k and 128 j/kg·k, respectively.
Answers: 1

Physics, 22.06.2019 20:50
An ideal otto cycle has a compression ratio of 8. at the beginning of the compression process, air is at 95 kpa and 27°c, and 750 kj/kg of heat is transferred to air during the constant-volume heat-addition process. assuming constant specific heats at room temperature, determine (a) the pressure and temperature at the end of the heat-addition process, (b) the net work output, (c) the thermal efficiency, and (d) the mean effective pressure for the cycle. (4390 kpa, 1730 k; 423 kj/kg; 56.4%; 534 kpa)
Answers: 1
You know the right answer?
Questions


Arts, 28.01.2021 01:00

Health, 28.01.2021 01:00



History, 28.01.2021 01:00




Mathematics, 28.01.2021 01:00


Biology, 28.01.2021 01:00


Mathematics, 28.01.2021 01:00

Mathematics, 28.01.2021 01:00

Mathematics, 28.01.2021 01:00


English, 28.01.2021 01:00

Mathematics, 28.01.2021 01:00