

Answers: 2


Another question on Physics


Physics, 22.06.2019 14:50
Nitrogen (n2) undergoes an internally reversible process from 6 bar, 247°c during which pν1.2 = constant. the initial volume is 0.1 m3 and the work for the process is 121.14 kj. assuming ideal gas behavior, and neglecting kinetic and potential energy effects, determine heat transfer, in kj, and the entropy change, in kj/s. show the process on a t-s diagram.
Answers: 2

Physics, 22.06.2019 15:00
Mechanical energy is when the amount of kinetic and potential energies added together remains the same. conserved created lost
Answers: 1

You know the right answer?
1. A soccer player runs directly up the field for 60 m before turning to the right at
an angle of 3...
Questions



Mathematics, 20.04.2021 16:10



Health, 20.04.2021 16:10

Mathematics, 20.04.2021 16:10

Mathematics, 20.04.2021 16:10


Computers and Technology, 20.04.2021 16:10



Mathematics, 20.04.2021 16:10



Business, 20.04.2021 16:10


Mathematics, 20.04.2021 16:10


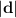 = √(10² + (60 + 10·√3)²) ≈ 77.96 m
= √(10² + (60 + 10·√3)²) ≈ 77.96 m


