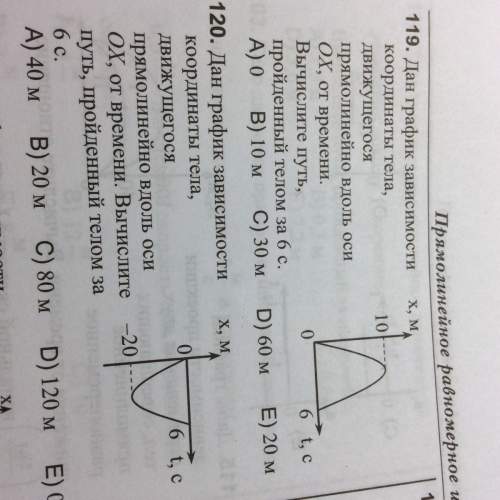Answers: 1


Another question on Physics

Physics, 22.06.2019 01:30
Ajet plane has a sound level of 120 db at a distance of 60 m. what is the q23. sound level at a distance of 6.0 km? (60)2 (6000)? om solution 80db db, hint: 16x assume ultrasound waves travel through the body of an animal at 1540 q24. m/s. if a 30,000 hz signal were reflected off a portion of a heart which was moving toward the source at 3 m/s, what frequency signal would return to the stationary source? solution 30.117hz
Answers: 3

Physics, 22.06.2019 02:00
In which situation has the independent variable for the experiment been changed? check all that apply. changed from counting the number of duckweed at day 0 and day 14 to counting them every day changed from testing duckweed growth in beakers to testing duckweed growth in large outdoor tanks changed from testing the effect of ph on duckweed growth to testing the effect of light levels on duckweed growth changed from testing the effect of acid ph on duckweed growth to testing the effect of basic ph on duckweed growth
Answers: 1

Physics, 22.06.2019 05:00
In a stagnant pool of water, a crayfish may spend much of its time lying with one side of its carapace near the surface of the water. in this position, it will move the walking legs on that side in a rhythmic back-and-forth motion. explain the likely function of this behavior.
Answers: 1

Physics, 22.06.2019 14:30
What distance does a car travel as its speed changes from 0 to 20 m/s in 17 s at constant acceleration?
Answers: 1
You know the right answer?
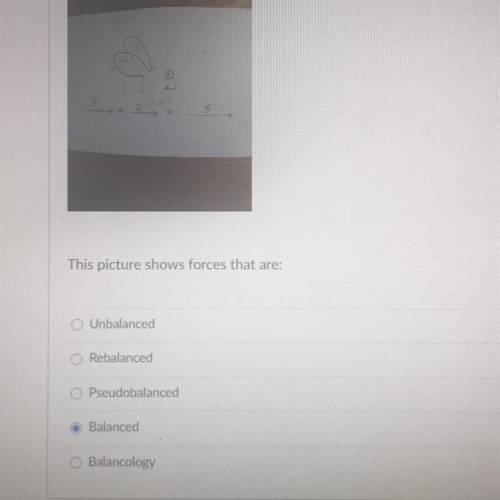
Please help. Hurry. Almost out of time. Hurry hurry.
...
...
Questions


Mathematics, 08.03.2021 17:30


History, 08.03.2021 17:30



Spanish, 08.03.2021 17:30

English, 08.03.2021 17:30

Mathematics, 08.03.2021 17:30



Mathematics, 08.03.2021 17:30

Mathematics, 08.03.2021 17:30

Mathematics, 08.03.2021 17:30

Computers and Technology, 08.03.2021 17:30

Arts, 08.03.2021 17:30

Geography, 08.03.2021 17:30


Physics, 08.03.2021 17:30