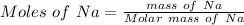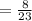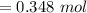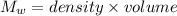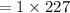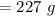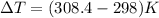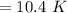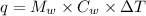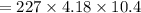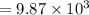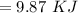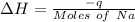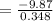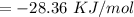Physics, 05.07.2020 14:01 flameftw2005
Find the enthalpy change per mole of sodium when sodium reacts with water. 8 grams of sodium reacts with 227 cm3 of water, producing a temperature change from 298 K to 308.4 K. The specific heat capacity of water is 4.18 J/K g. A. -28.36 kJ B. 9868.1 J C. 62.75 kJ D. -28356.7 kJ

Answers: 1


Another question on Physics

Physics, 22.06.2019 05:40
Karen is running forward at a speed of 9 m/s. she tosses her sweaty headband backward at a speed of 20 m/s. the speed of the headband, to the nearest whole number relative to a stationary observer watching karen
Answers: 1

Physics, 22.06.2019 11:30
Write down two additional questions about earth's magnetic field that will make your presentation more informative.
Answers: 1

Physics, 22.06.2019 19:30
Water is siphoned from a large tank and discharges into the atmosphere through a 50-mm diameter tube. the end of the tube is b = 2.6 m below the tank bottom which is a = 6.7 m deep, and viscous effects are negligible. determine the maximum height h over which the water can be siphoned without cavitation occurring. atmospheric pressure is 101.4 kpa, and the water vapor pressure is 1.79 kpa (absolute). report your answer in meters to two decimal places.
Answers: 1

You know the right answer?
Find the enthalpy change per mole of sodium when sodium reacts with water. 8 grams of sodium reacts...
Questions

Mathematics, 13.04.2021 19:00

Mathematics, 13.04.2021 19:00



History, 13.04.2021 19:00



Mathematics, 13.04.2021 19:00

Mathematics, 13.04.2021 19:00

Chemistry, 13.04.2021 19:00


Mathematics, 13.04.2021 19:00


Mathematics, 13.04.2021 19:00

Spanish, 13.04.2021 19:00


Mathematics, 13.04.2021 19:00

Mathematics, 13.04.2021 19:00

Mathematics, 13.04.2021 19:00

Chemistry, 13.04.2021 19:00

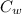 ) = 4.18 J/Kg
) = 4.18 J/Kg