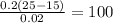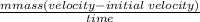A bowling ball has a momentum of 240 kgm/s
...

Answers: 2


Another question on Physics

Physics, 22.06.2019 00:10
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3

Physics, 22.06.2019 12:10
Energy flows from the producer level to the level. is called
Answers: 1

Physics, 22.06.2019 20:50
Asquare 1m x 1m plate is shown below. all four of its edges are kept at 0 co. at t=0the temperature distribution is given by: y xtsins n75)=.a) using separation of variables determine the temperature distribution t(x,y,t)along the plate at any time t.b) discretize the heat conduction equation +=22222 txtusing a second-order centered difference scheme for the two second derivatives and a first-order forward euler scheme for the first derivative. using von neumann stability analysis, determine the stability criterion for the proposed discretization scheme in terms of , hand t. solve for the maximum allowable time step tmax
Answers: 1

Physics, 23.06.2019 02:30
Substances that assume the shape of their container, but do not have a definite size are called
Answers: 2
You know the right answer?
Questions

Biology, 19.07.2019 07:00





Social Studies, 19.07.2019 07:00

Biology, 19.07.2019 07:00










Chemistry, 19.07.2019 07:00

Mathematics, 19.07.2019 07:00

Physics, 19.07.2019 07:00

Health, 19.07.2019 07:00