
Physics, 27.03.2020 05:16 Lovemimi4129
Using the following equation for the combustion of octane, calculate the heat associated combustion of 100.0 g of octane assuming complete combustion. The molar mass of octane is 114.33 g/mole. The molar mass of oxygen is 31.9988 g/mole.2C8H18+25O2→16CO2+18H2O;ΔH∘r xn = -11018 kJA. -11018 kJB. -4819 kJC. -602.3 kJD. -535.4 kJE. -385.5 kJ

Answers: 2


Another question on Physics

Physics, 21.06.2019 12:30
11 words used to describe an object are called itsor characteristics.total12 is the amount of matter in an object.helt udedelerde elettroduodet littletadli dituntut uploaded13 the metric unit used to measure weight is them.14 if a toy has a mass of 1 kg, it has an earth weight 15 the density of liquid water isti
Answers: 1

Physics, 22.06.2019 17:30
The items in the following list are all units of matter. which is the smallest unit that retains the properties of the matter? a.) atom b.) compound c.) electron d.) element
Answers: 1

Physics, 22.06.2019 18:30
What would people living along the coast in south florida do if there was a hurricane warning? move to locations away from the water flock along coasts to watch the natural phenomenon buy instruments to predict the exact location of the hurricane measure water levels to know the exact time of the hurricane
Answers: 3

Physics, 22.06.2019 19:40
It may seem strange that the selected velocity does not depend on either the mass or the charge of the particle. (for example, would the velocity of a neutral particle be selected by passage through this device? ) the explanation of this is that the mass and the charge control the resolution of the device--particles with the wrong velocity will be accelerated away from the straight line and will not pass through the exit slit. if the acceleration depends strongly on the velocity, then particles with just slightly wrong velocities will feel a substantial transverse acceleration and will not exit the selector. because the acc
Answers: 1
You know the right answer?
Using the following equation for the combustion of octane, calculate the heat associated combustion...
Questions


Biology, 28.07.2019 20:30


English, 28.07.2019 20:30





Business, 28.07.2019 20:30


Business, 28.07.2019 20:30


Mathematics, 28.07.2019 20:30


English, 28.07.2019 20:30

Mathematics, 28.07.2019 20:30

History, 28.07.2019 20:30

English, 28.07.2019 20:30

Mathematics, 28.07.2019 20:30

Mathematics, 28.07.2019 20:30

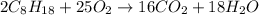 Δ
Δ = -11018 kJ
= -11018 kJ

