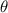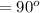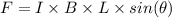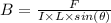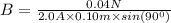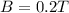Answers: 1


Another question on Physics

Physics, 22.06.2019 14:50
Nitrogen (n2) undergoes an internally reversible process from 6 bar, 247°c during which pν1.2 = constant. the initial volume is 0.1 m3 and the work for the process is 121.14 kj. assuming ideal gas behavior, and neglecting kinetic and potential energy effects, determine heat transfer, in kj, and the entropy change, in kj/s. show the process on a t-s diagram.
Answers: 2

Physics, 22.06.2019 16:30
Humidity is to blame for that muggy, steamy feeling you experience on some hot summer days. what gas in the atmosphere causes humidity? a) oxygen b) hydrogen c) nitrogen d) water vapor
Answers: 1

Physics, 23.06.2019 01:00
What will happen in a hybrid vehicle if current flow becomes too high due to a short?
Answers: 1

Physics, 23.06.2019 09:50
How much heat is needed to raise the temperature of 5g of water by 16°c?
Answers: 2
You know the right answer?
A straight wire 0.10 m long carrying a current of 2.0 A is at right angles to a magnetic field. The...
Questions

Mathematics, 07.03.2020 01:37

English, 07.03.2020 01:37

Mathematics, 07.03.2020 01:37



Mathematics, 07.03.2020 01:37




Mathematics, 07.03.2020 01:37

English, 07.03.2020 01:37

Mathematics, 07.03.2020 01:37






Computers and Technology, 07.03.2020 01:37