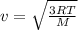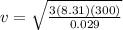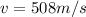Physics, 28.11.2019 18:31 keiannabmoss
The temperature of an ideal gas is directly proportional to the average kinetic energy of its molecules. if a container of an ideal gas is moving past you at 2000 m/s, is the temperature of the gas higher than if the container was at rest? explain your reasoning.

Answers: 2


Another question on Physics

Physics, 22.06.2019 15:20
Abag of potato chips contains 2.00 l of air when it is sealed at sea level at a pressure of 1.00 atm and a temperature of 20.0 deg c. what will be the volume of the air in the bag if you take it with you, still sealed, to the mountains where the temperature is 7.00 deg c and atmospheric pressure is 70.0 kpa? assume that the bag behaves like a balloon and that the air in the bag is in thermal equilibrium with the outside air.
Answers: 3

Physics, 23.06.2019 00:20
The brightest star in the night sky in the northern hemisphere is sirius. its distance from earth is estimated to be 8.7 light years. a light year is the distance light travels in one year. light travels at a speed of 3.00 × 108 m/s. calculate the distance from earth to sirius in miles. (1 mi = 5280 ft) g
Answers: 2

Physics, 23.06.2019 00:30
What is the relationship between globular clusters and the milky way?
Answers: 1

Physics, 23.06.2019 01:20
Afisherman notices that his boat is moving up and down periodically, owing to waves on the surface of the water. it takes a time of 2.70 s for the boat to travel from its highest point to its lowest, a total distance of 0.700 m. the fisherman sees that the wave crests are spaced a horizontal distance of 6.50 m apart. a. how fast are the waves traveling? b. what is the amplitude a of each wave?
Answers: 1
You know the right answer?
The temperature of an ideal gas is directly proportional to the average kinetic energy of its molecu...
Questions

Biology, 16.07.2019 08:20

History, 16.07.2019 08:20

Social Studies, 16.07.2019 08:20

History, 16.07.2019 08:20

Biology, 16.07.2019 08:20


Social Studies, 16.07.2019 08:20


Mathematics, 16.07.2019 08:20




Computers and Technology, 16.07.2019 08:20

English, 16.07.2019 08:20

Spanish, 16.07.2019 08:20