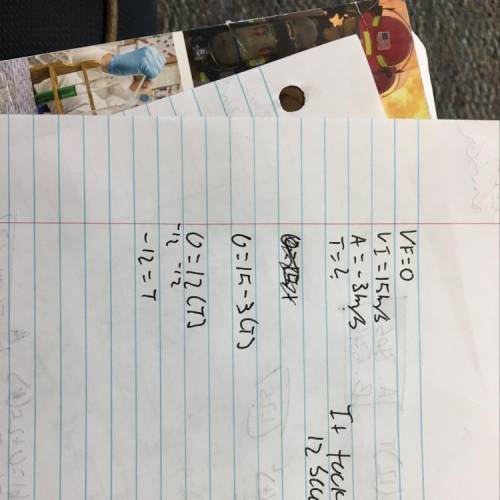Loads of points - show working and method used
a cyclist is traveling at 15 m/s and app...


Answers: 1


Another question on Physics

Physics, 21.06.2019 17:00
These models show the electron structures of two different nonmetal elements. which element is likely more reactive, and why? element 1 is more reactive because it has fewer electron shells and is toward the top of its group on the periodic table. element 1 is more reactive because it has more electrons in its valence shell and is farther to the right on the periodic table. element 2 is more reactive because it does not have a valence shell close to the nucleus, so it will attract electrons. element 2 is more reactive because it does not have a full valence shell, so it will attract electrons.
Answers: 2


Physics, 22.06.2019 16:30
Iron is a transition metal with multiple oxidation numbers. (answer the following) a. what is the iron (ii) ion? how does it differ from the iron (iii) ion? b. if iron were to bond with oxygen, predict the formula for each oxidation number of iron. c. how would each formula be named?
Answers: 2

You know the right answer?
Questions

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Social Studies, 11.09.2020 09:01

English, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Biology, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

English, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

English, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01

Mathematics, 11.09.2020 09:01