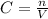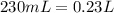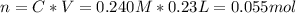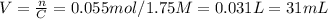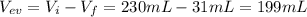Physics, 10.09.2019 21:20 msjsnell29
A230.-ml sample of a 0.240 m solution is left on a hot plate overnight; the following morning, the solution is 1.75 m. what volume of water evaporated from the 0.24 m solution?

Answers: 1


Another question on Physics

Physics, 22.06.2019 04:00
Determine the maximum r-value of the polar equation r =3+3 cos 0
Answers: 3

Physics, 22.06.2019 07:10
1. how much energy is needed to raise the temperature of 40.0 g of argon from 25c to 40c? the specific heat capacity of argon is 0.520 j/(g·k) 2a. 23.0 ml of 0.100 m hcl (standard) are added from a buret to neutralize 50.0 ml of an unknown basic solution. 2b. if the oh- produced in the previous reaction came from ca(oh)2, then what is the molarity of the ca(oh)2? 3.calculate the new freezing-point of a solution when 60.5 grams of cacl2 solute is dissolved in 0.612 kg of water. 4. what is the maximum number of moles of alcl3 that can be produced from 5.0 mol al and 6.0 mol cl2? 5. a sample of oxygen gas has a volume of 150 ml when its pressure is 0.923 atm. if the pressure is increased to 0.987 atm and the temperature remains constant, what will the new volume be? 6. nitrogen gas in a closed container at a temperature of 100.0 oc and 3.0 atm is heated to 300 oc. what is the pressure of the gas at the higher temperature?
Answers: 3

Physics, 22.06.2019 22:50
Which of the following statements correctly describes a transformer?
Answers: 3

Physics, 23.06.2019 01:30
Which statements did both aristotle and ptolemy assume? check all that apply.
Answers: 1
You know the right answer?
A230.-ml sample of a 0.240 m solution is left on a hot plate overnight; the following morning, the...
Questions



Social Studies, 09.11.2020 03:20

Mathematics, 09.11.2020 03:20


Chemistry, 09.11.2020 03:20


Mathematics, 09.11.2020 03:20



Mathematics, 09.11.2020 03:20

Chemistry, 09.11.2020 03:20


Computers and Technology, 09.11.2020 03:30

Health, 09.11.2020 03:30


Computers and Technology, 09.11.2020 03:30

English, 09.11.2020 03:30